Get a Free WorkTrek Demo
Let's show you how WorkTrek can help you optimize your maintenance operation.
Try for freeKey Takeaways:
- Equipment validation is a formal, documented process that confirms your equipment performs reliably and consistently within predefined acceptance criteria.
- The validation process follows three core qualification stages: Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ).
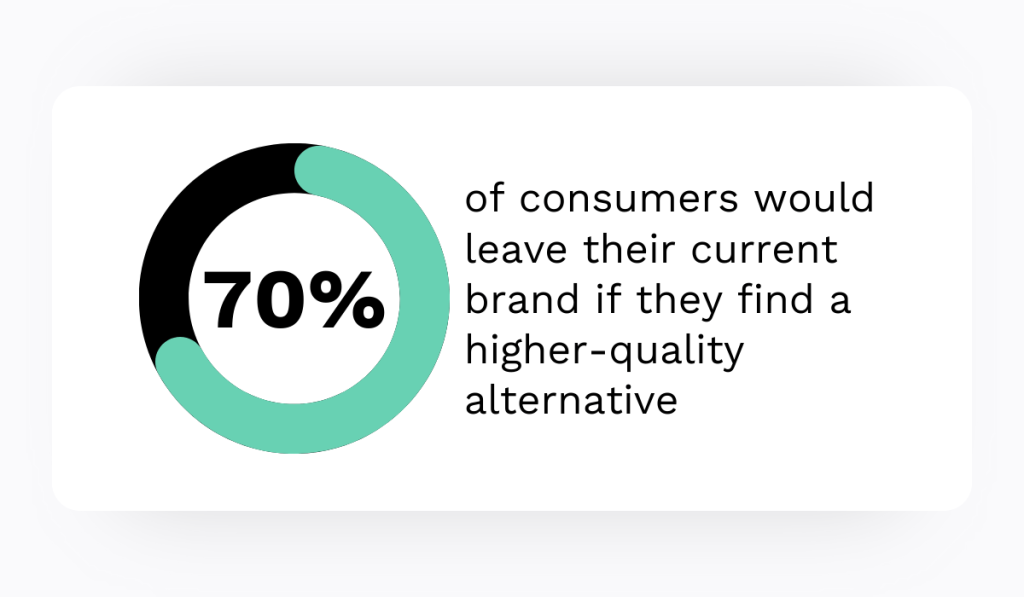
- In 2024, the EMA conducted 210 GMP inspections for centrally authorized medicines, with 10 leading to non-compliance statements that blocked EU supply.
- A CMMS like WorkTrek helps manufacturers maintain validated equipment, store documentation, and stay inspection-ready at all times.
Regulatory bodies do not accept good intentions as evidence of compliance. They want documented proof.
If your manufacturing equipment cannot demonstrate that it performs reliably, within specified operational parameters, and in accordance with predefined acceptance criteria, you are exposed to warning letters, costly recalls, and potential production shutdowns.
Equipment validation is the systematic process that provides proof. It is how regulated industries confirm that all the equipment involved in production processes meets the necessary quality standards before it is ever used to produce a finished product.
This guide covers everything you need to know.
From understanding what equipment validation is and why it matters, to the key phases, regulatory requirements, and how a CMMS can help your team maintain a validated state long after the initial qualification is complete.
What Is Equipment Validation?
Equipment validation is the process of collecting and evaluating documented evidence that a piece of equipment consistently performs as intended and in accordance with its manufacturer’s specifications.
It is a core requirement under Good Manufacturing Practice (GMP) regulations and quality management systems such as ISO 13485 and ISO 9001. It also forms a central part of any validation master plan.
The goal is straightforward: ensure consistent quality across every production run. That means your pharmaceutical, laboratory, and analytical equipment, as well as critical instruments, all need to be verified and documented before they are used in regulated production processes.
Equipment validation is not a one-time event. It is an ongoing commitment.
Any time equipment is modified, relocated, or repaired, revalidation activities are required to confirm that the equipment still meets its critical equipment parameters.
Equipment validation is required when:
- New equipment is installed and prepared for use
- Existing equipment is modified or undergoes significant repairs
- Manufacturing processes change or new raw materials are introduced
- Periodic regulatory reviews or internal quality audits are triggered
- Equipment fails or shows performance outside its operational parameters
Why Equipment Validation Matters
The consequences of skipping or poorly executing validation activities are severe.
In 2024, the EMA reported 210 GMP inspections for centrally authorized medicines, with 10 inspections leading to non-compliance statements. Those outcomes blocked EU supply for the affected products.
In the United States, the FDA cited a drug manufacturer in 2024 for failing to conduct adequate cleaning validation studies for multipurpose equipment and ordered the company to cease all manufacturing operations until the issue was resolved.
Beyond regulatory penalties, equipment that operates outside its validated state can compromise final product quality and, in pharmaceutical manufacturing and medical devices, directly threaten patient safety.
More than 60% of regulatory observations in pharmaceutical manufacturing relate to documentation control, investigation quality, and procedural compliance gaps. Robust equipment validation, properly documented, is one of the most direct ways to close those gaps.
Equipment validation matters because it protects three things at once: your patients, your products, and your business.
The Three Core Phases: IQ, OQ, PQ
The equipment qualification process follows a structured lifecycle. Each phase builds on the previous one and is supported by validation protocols, documented test procedures, and clear acceptance criteria.
1. Installation Qualification (IQ)
Installation qualification confirms that the equipment has been delivered, installed, and configured correctly in accordance with manufacturer specifications and user requirements.
During IQ, the validation team verifies that critical components are present and intact, that utilities are connected properly, and that the equipment installation matches the approved design specifications.
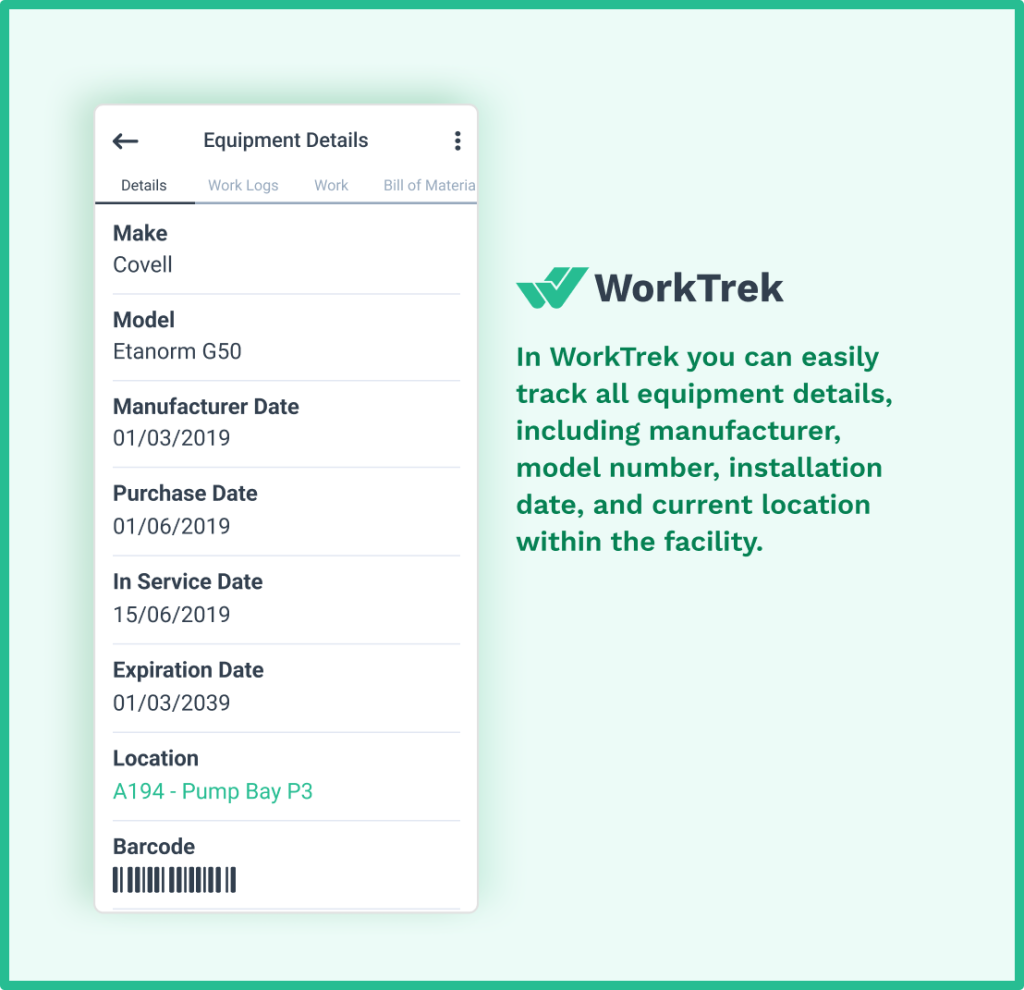
IQ documentation typically covers:
- Equipment model, serial numbers, and delivery records
- Confirmation against the manufacturer’s specifications and functional specifications
- Utility connections, including electrical, water, and gas
- Instrument calibration records for critical instruments
- Safety procedures and operator training requirements
2. Operational Qualification (OQ)
Operational qualification (OQ) confirms that the equipment operates as intended across its full range of operating procedures. The validation team runs the equipment through defined test conditions to verify that it consistently meets its operational parameters.

OQ challenges the equipment at the boundary conditions.
This means testing not just at typical operating settings, but at the upper and lower limits of the equipment’s specified range to confirm it performs reliably throughout.
OQ activities include:
- Verifying critical process parameters against predefined acceptance criteria
- Testing alarm systems, interlocks, and safety controls
- Confirming that standard operating procedures (SOPs) are adequate
- Documenting all test procedures and deviations with corrective actions
3. Performance Qualification (PQ)
Performance qualification (PQ) is the final stage of the IQ OQ PQ sequence. It confirms that the equipment performs reliably under actual production conditions, using real materials and following approved operating procedures.

Where OQ tests the equipment in isolation, PQ tests it in context.
The equipment must demonstrate consistent quality output across multiple production runs, with results that meet predefined acceptance criteria every time.
Successful PQ completion means the equipment is formally in a validated state and cleared for use in regulated manufacturing processes.
Process Validation vs. Equipment Qualification
These two terms are closely related but address different things.
Equipment qualification, covering IQ, OQ, and PQ, focuses on the individual piece of equipment. It confirms that the equipment functions correctly, safely, and consistently within its operational parameters.
Process validation focuses on the entire manufacturing process.
It confirms that the production process as a whole reliably produces a high-quality product that meets its specifications. This includes evaluating raw materials, production parameters, and final product quality.
Both are required for full regulatory compliance. Equipment qualification provides the foundation. Process validation builds on top of it.
The FDA’s guidance on Process Validation: General Principles and Practices outlines a three-stage lifecycle approach: process design, process qualification, and continued process verification. Each stage relies on validated equipment as a prerequisite.
Key Components of an Equipment Validation Protocol
A well-structured validation protocol is the foundation of every successful validation project. It serves as the official roadmap that the validation team follows from start to finish.
Every equipment validation protocol should include:
- Scope and purpose: what the protocol covers and why it is being conducted
- User requirement specifications (URS): what the end user needs the equipment to do
- Functional specifications: how the equipment will achieve those requirements
- Risk assessment: identifying critical components, critical process parameters, and failure modes
- Test procedures: step-by-step instructions for each qualification activity
- Predefined acceptance criteria: the measurable thresholds each test must meet to pass
- Traceability matrix: mapping each requirement to a specific test
- Documentation requirements: what records must be created and retained as documented evidence
- Deviation procedures: how to handle results that fall outside acceptance criteria

Skipping any of these components creates gaps that inspectors will find. A complete, accurate protocol also makes future validation efforts faster because the framework is already in place.
Regulatory Requirements and Standards
Equipment validation guidelines vary by industry and geography, but the core expectations from regulatory bodies are consistent.
Key regulatory frameworks include:
- FDA 21 CFR Parts 210 and 211: Good Manufacturing Practice regulations for pharmaceutical manufacturing in the United States
- EU GMP Annex 15: The European Commission’s guidelines on qualification and validation, covering the full equipment lifecycle
- ISO 13485: Quality management systems requirements for medical devices, including equipment validation provisions
- ICH Q9: Quality Risk Management guidance, which informs the risk assessment approach within validation activities
- WHO TRS guidelines: World Health Organization technical reports that define validation expectations for global pharmaceutical manufacturing
Regulatory inspection readiness is one of the most practical drivers of consistent validation practice. Inspectors want to see documented evidence, not verbal assurances.
Data integrity is a particular area of focus. Every piece of documented evidence must be attributable, legible, contemporaneous, original, and accurate. These are the ALCOA principles that underpin any audit-ready quality system.
Equipment Validation Best Practices
Knowing the steps is not enough. Execution quality is what separates teams that pass inspections from those that do not.
1. Conduct a thorough risk assessment before starting.
Not all equipment carries the same level of risk. Critical equipment that directly creates a product or controls critical process parameters requires more rigorous validation than non-critical support equipment. Use a risk-based approach to prioritize your validation activities and allocate resources accordingly.

2. Build your traceability matrix early.
A traceability matrix connects every user requirement specification to a specific test. It confirms that nothing has been overlooked and that all the equipment functions intended by the user have been tested against predefined acceptance criteria.
3. Document everything in real time.
Documented evidence recorded after the fact raises questions about data integrity. Maintain maintenance records, calibration logs, and qualification reports as the work happens.
4. Train your validation team thoroughly.
The people executing validation protocols must have a thorough understanding of both the equipment and the regulatory requirements. A comprehensive qualification effort falls apart if the team executing it does not know what they are looking for. Focus on training your team.
5. Plan for revalidation from day one.
Validated equipment needs ongoing management. Build revalidation triggers into your operating procedures, including after maintenance, process changes, or equipment moves. This protects your validated state and supports continuous improvement.
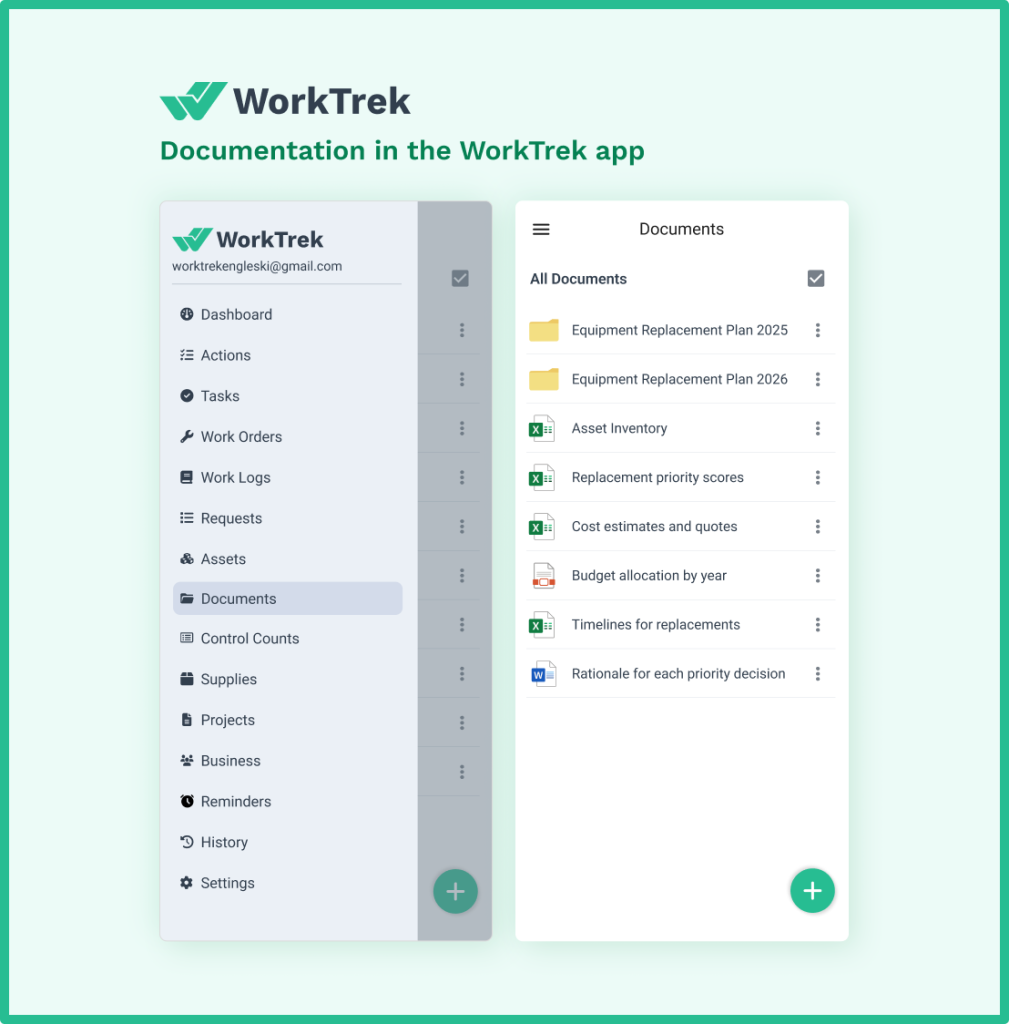
How a CMMS Supports Equipment Validation
Most teams struggle with equipment validation, not because they lack knowledge, but because they lack the systems to manage it at scale.
Maintenance records get scattered. Calibration schedules are missed. Validation documents live in binders that no one can find during an inspection. These are not hypothetical problems. They are the most common reasons that facilities fail audits.
A Computerized Maintenance Management System (CMMS) directly addresses all of these issues.
Industries That Require Equipment Validation
Equipment validation is not exclusive to the pharmaceutical industry, though pharmaceutical equipment validation and pharmaceutical manufacturing remain the most visible context due to stringent FDA and EMA oversight.
Regulated industries that require formal validation include:
- Pharmaceutical manufacturing: drug production, formulation, and packaging require validated equipment at every stage
- Medical devices: ISO 13485 and FDA 21 CFR Part 820 mandate equipment qualification for device production
- Biotechnology: production of biologics and cell-based therapies involves highly specialized, validated equipment
- Food and beverage: equipment used in critical processing steps must meet safety and quality standards
- Aerospace: equipment used to manufacture safety-critical components requires formal qualification
- Laboratory and analytical equipment: any instrument used to generate data that informs product release decisions requires validation
In each of these sectors, the principle is the same: equipment that is not validated is a liability.
The level of formality and regulatory scrutiny varies, but the need for documented evidence that equipment performs reliably does not.
Common Pitfalls to Avoid
Even experienced teams make validation mistakes. Knowing where others have gone wrong helps you avoid the same problems.
The most common pitfalls include:
- Incomplete user requirement specifications: when URS documents are vague, it becomes impossible to write meaningful acceptance criteria or test procedures
- Poor change control: modifications to equipment or production processes without triggering revalidation are among the most cited deficiencies during inspections
- Gaps in maintenance records: validation establishes a baseline, but if maintenance records do not demonstrate that the equipment has been kept in its qualified condition, that baseline becomes meaningless
- Treating validation as a one-time project: equipment qualification is not a checkbox. It is a continuous quality management commitment
- Relying on manual documentation: paper-based systems create data integrity risks and make inspection preparation extremely time-consuming
These pitfalls are not inevitable.
With the right maintenance strategy and the right tools in place, they are entirely preventable.
Conclusion
Equipment validation is a critical function in any regulated manufacturing environment. It confirms that your equipment performs reliably, that your production processes consistently deliver high-quality products, and that your organization can demonstrate compliance at any time.
The IQ OQ PQ framework gives you a structured, repeatable pathway from equipment installation to full production readiness. But the work does not stop at PQ sign-off.
Maintaining validated equipment requires ongoing discipline: regular calibration, documented maintenance records, and a clear process for managing changes.
Teams that treat validation as a continuous quality management activity, supported by strong systems and clear procedures, are the ones that pass inspections, avoid recalls, and consistently deliver safe, effective products to patients and customers.